You may find this question familiar, or have had head-scratching moments pondering over the answers given to your child — where does the heat go to after it has been conducted away by the water?
Balloon A is filled with air only, while Balloon B is filled with air and a little water. Balloon A and Balloon B are each positioned above a flame. Balloon A bursts first, followed a little while later by Balloon B.
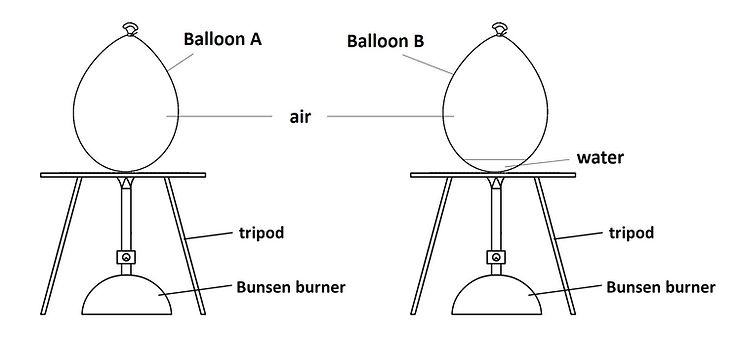
The question: why does Balloon B burst later?
First, we’ll need to identify what actually causes the rubber balloon to burst in both cases. There are 2 suspects:
(a) expansion of air inside the balloon which causes the balloon to swell until it bursts, and
(b) expansion of the rubber at the spot where heat is applied.
For (a), the balloon bursts due to uniform expansion of the entire balloon (since air is pushed out in all directions); while for (b), the balloon bursts due to the stretching of the balloon at a small area (i.e. non-uniform expansion of the balloon).
Since rubber is a poor conductor of heat, it turns out (b) is the cause of the balloon ‘going out with a bang’. In other words, the temperature of the balloon (at the spot where heat is applied) has reached the critical temperature where it starts to tear.
Now, the next question: how does water slow down the rubber’s increase in temperature?
Well, the answer is straightforward in this case. Water simply conducts the heat away. Compared to air, water is a better conductor of heat; the water in Balloon B removes heat from the rubber more quickly than the air in Balloon A does. Thus, for Balloon B, the rubber takes a longer time to reach that critical temperature, let’s call it C, where it starts to tear. Just as importantly, water is able to absorb all that heat without as great a rise in temperature as compared to air, but more on that later.
There is, however, a variant of the question circulating in the local primary science circuit that is a little more befuddling.
In this variant, Balloon A and Balloon B are now filled with equal volumes of different unidentified substances — X and Y — respectively. Again, Balloon A bursts first.

In this case, can we apply the same logic and say for sure that substance X is a poorer conductor of heat than substance Y? Well, not really …
Not knowing what X and Y are makes this question a little more complicated because the concept of specific heat capacity (the amount of heat energy required to raise the temperature of a unit mass of a substance by 1 degree Celsius) comes into play.
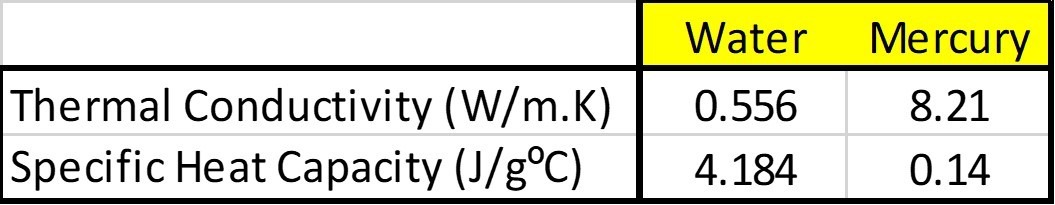
Let’s assume X is mercury and Y is water. Here’s how they stack up against each other.

In layman terms, this means mercury conducts heat 15 times more quickly than water but requires 30 times less heat energy than water to raise its temperature by 1 degree Celsius for the same unit mass.
So, while mercury can conduct heat away from the rubber more quickly compared to water, its temperature also starts to rise more quickly. This effect becomes more pronounced if the mass of mercury is small, since its capacity to absorb heat will be limited. What this means is that Balloon A will burst first even though it contains the better heat conductor!
Why was this not a consideration for the first question? It’s because, when compared to air, water not only conducts away heat more quickly, it also requires more heat for the same increase in temperature per unit mass.

From the diagram, it is the volumes of substances X and Y that are equal, not their masses. And because mercury has a higher density than water, mercury will have a greater mass than water for the same volume…which adds another level of complexity!
So, to conclude that Y is definitely a better heat conductor than X because Balloon A bursts first is scientifically hasty. As we’ve shown, that might hold true when X is mercury and Y is water!
Click the share button below if you found this useful and subscribe to our newsletter to receive your regular dose of PSLE Tip Of The Week! (#PSLE_TOTW)